 The Lewis structure of Silicon tetrafluoride gives us information that the silicon atom forms four single bonds with four fluorine atoms. Hence, the most suitable Lewis structure of silicon fluoride in bond notation form would be: Two electrons shared between the silicon and fluorine atom form a single bond in silicon tetrafluoride. Therefore, the silicon and fluorine atoms do not carry any charge in the Lewis structure of silicon tetrafluoride. The formal charge on the Fluorine atom, F = 7 −12(2) − 6 = 0 The formal charge on the Silicon atom, Si = 4 −12(8) − 0 = 0 Now, let us calculate the formal charge on the silicon and fluorine atoms in SiF4, which is defined as:įormal charge = Number of valence electrons − 1/2(bonding electrons) – nonbonding electrons We can observe that both silicon and fluorine have completed their octet i.e., they have eight electrons in their outermost shell. 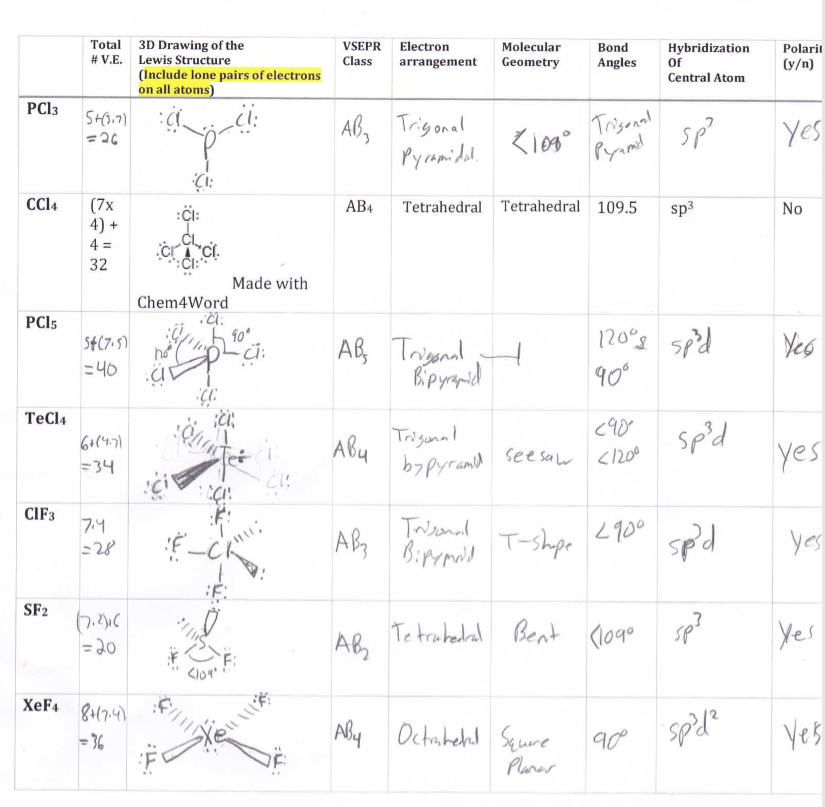
Here, we have arranged the 32 valence electrons in silicon tetrafluoride, as shown in the following diagram: Octet Rule: Every element in the periodic table (except Hydrogen and Helium atom) tend to have a noble gas configuration during the formation of a chemical bond with another atom. Step 3: Organization of all valence electrons in silicon tetrafluoride by following the octet rule. The four fluorine atoms will surround the silicon atom. Out of silicon and fluorine atoms, silicon is less electronegative than fluorine, which makes it a central atom in silicon tetrafluoride. Step 2: Find out the central atom in silicon tetrafluoride. Therefore, silicon tetrafluoride consists of 4 + (7*4) = 32 valence electrons in total. Hence, the silicon atom has four valence electrons whereas the fluorine atom has seven valence electrons. The silicon and fluorine atoms belong to group 14, and group 17, respectively. Silicon tetrafluoride contains one silicon atom and four fluorine atoms. Step 1: Find out the total number of valence electrons in silicon tetrafluoride. Let us draw the Lewis structure of silicon tetrafluoride. These valence electrons or outermost shell electrons of the atoms are represented as dots and hence, it is also known as electron dot structure. The Lewis structure refers to the two-dimensional representation of the molecule with valence electrons surrounding the atoms in the molecule. 
Then, we will study the polarity of SiF4 i.e., whether SiF4 is a polar or nonpolar molecule.įirst of all, there is a need to understand the Lewis structure of silicon tetrafluoride for studying its chemical bonding. In this article, we are going to discuss the chemical bonding of silicon tetrafluoride by understanding its Lewis structure, molecular geometry, and the hybridization of the central atom. The density of SiF4 is 1.66 g/cm3, which makes it heavier than air. The volatile nature of silicon tetrafluoride limits their use and hence, it is restrained to only organic synthesis and microelectronic. 
Silicon tetrafluoride is prepared in the laboratory by decomposition of BaSiF6 at a temperature greater than 300 ☌.īaSiF6 -–> SiF4 (volatile) + BaF2 (residue) Silicon tetrafluoride is a colorless, toxic, corrosive, and non-flammable gas with a pungent smell. The melting and boiling point of silicon tetrafluoride is -95.0 ☌ and -90.3 ☌ and hence, it exists as a gas at room temperature. It is named tetrafluorosilane or silicon tetrafluoride. Electronegative ligands such as F will always go to the axial sites.SIF4 is a covalent compound, which consists of silicon and fluorine atoms. In general, by this reasoning, lone pairs and electropositive ligands such as CH 3 will always prefer the equatorial sites in the trigonal bipyramidal geometry. = 0.867 \:bond (formal \: charge = -0.122)\)īecause fluorine is more electronegative than a lone pair, it prefers the axial site where it will have more negative formal charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
